Biological Indicators for Sterilization
Biological Indicators for Sterilization
-
Sterilization & Cleaning Monitoring
Elevate Sterilization Confidence with Mesa Labs Biological Indicators and Chemical Indicators
Known globally for our experience and expertise, Mesa Labs manufactures AAMI-, ISO-, and USP-compliant biological indicator and chemical indicators for a wide range of sterilization modalities. Our products and custom solutions offer convenient and consistent ways to validate and monitor the compliance of your processes and products.
Indicators by process
Mesa Labs Biological and Chemical indicators support an array of sterilization processes, and include strips, discs, ampoules, self-contained, suspension, UniQ, and custom options.
Elevate your sterilization & disinfection control
We’re known for our consultative approach, applying deep expertise to meet your most critical needs: cycle development, validation, and monitoring; standard, UniQ, and custom indicators; and full-service support.
Quality
Our standard and UniQ indicators are manufactured in an ISO-certified facility to the most exacting specifications. Our quality earns customer trust for BI manufacturing worldwide.
Expertise
Mesa provides in-depth technical support and consulting at every stage. Whatever your application demands, we give you access to advanced insights and experience.
Variety
Mesa’s broad portfolio matches a wide variety of sterilization processes. We work with you to identify the most appropriate standard and specialized solutions to meet your needs.
Industries
BIOPHARMACEUTICAL
MED DEVICE
HEALTHCARE
FOOD & BEVERAGE
Get the inside edge
Full of expert takes on industry trends and the latest technical education offerings, Spore News gives you the insight you need to prepare for what’s next.
COMPLIANCE
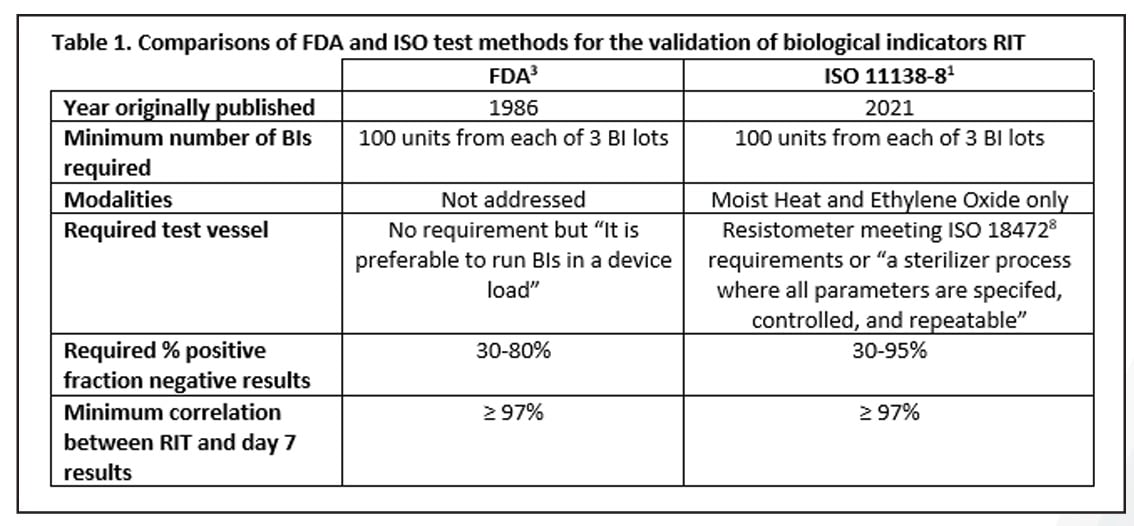
A Review of the Newly Released ISO 11138-8 Standard on the Validation of a Reduced Incubation Time for Biological Indicators
Read Mesa Labs' Spore News to learn about the newly developed ISO standard for the validation of a reduced incubation time as part of the 11138 series.

D-VALUE STUDIES
Product D-Value Study Part 2, Heat-sensitive & Atypical Aqueous Liquids
Read Mesa Labs' Spore News to learn about the sterilization and microbiological validation of heat sensitive products.

D-VALUE STUDIES
Why Perform a D-value Study?
D-value studies are discussed in detail in the Spore News publications referenced below.

STEAM-IN PLACE
The MeCo Solution: Improving Consistency, Accuracy and Robustness in SIP Validation
Read Mesa Labs’ Spore News to learn about how MeCo improves consistency, accuracy and robustness in sterilization in place (SIP) validation.

IDENTIFICATION
Sub-culturing a Positive Biological Indicator for Identification
Read Mesa Labs’ Spore News to learn about sub-culturing procedures to identify organisms responsible for positive biological indicators.
COMPLIANCE
Biological Indicators and the European Pharmacopoeia
Read Mesa Labs’ Spore News to learn about biological indicators and the European Pharmacopoeia 9.2, 5.1.2.
CATEGORY
Lorem ipsum dolor sit amet, consectetur adipiscing elit.
Pellentesque rutrum purus nec posuere ultricies.