Process Challenge Devices® (PCD®)
-
Process Challenge Devices® (PCD®)
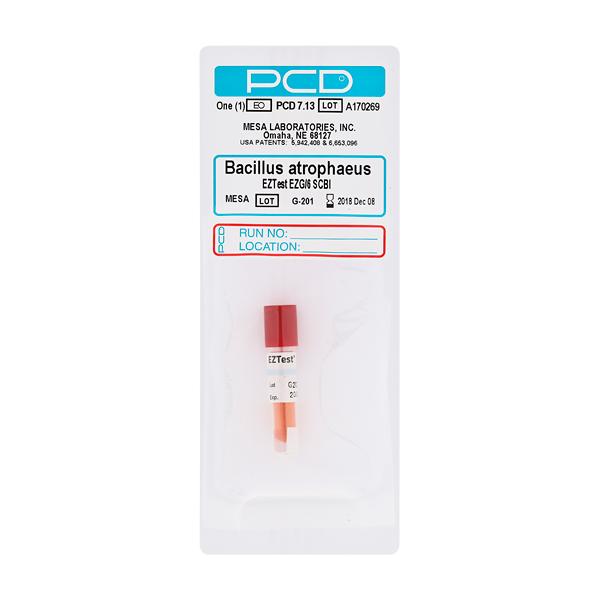
Process Challenge Devices® (PCD®) are externally placed on pallets to assure terminal sterilization of medical devices. PCDs have defined resistance values that match internal sterilization challenges.
- Mesa PCD® have unique patented packaging of a variety of B. atrophaeus biological indicators to match your process.
- You can use PCD® for both validation and routine monitoring of Ethylene Oxide sterilization cycles.
- PCDs have consistent resistance and are certified to ISO 13485 quality systems for medical devices.
- Mesa’ s PCD® line offers a broad range of available resistance levels and biological indicator (BI) types to meet endless cycle configurations. See the product section below…
- Our PCD® Selection Set offers 4 of the most common configurations in one kit to determine appropriate resistance level for your cycle.
- We can provide faster turnaround with self-contained biological indicators.
Products
Type 2 (Most Resistance)
PCD® 2.1 Mesa BI Disc, 1-10006mm (D-Value: 28)
PCD® 2.12 Mesa BI Strip, SGMG/6 (D-Value: 28)
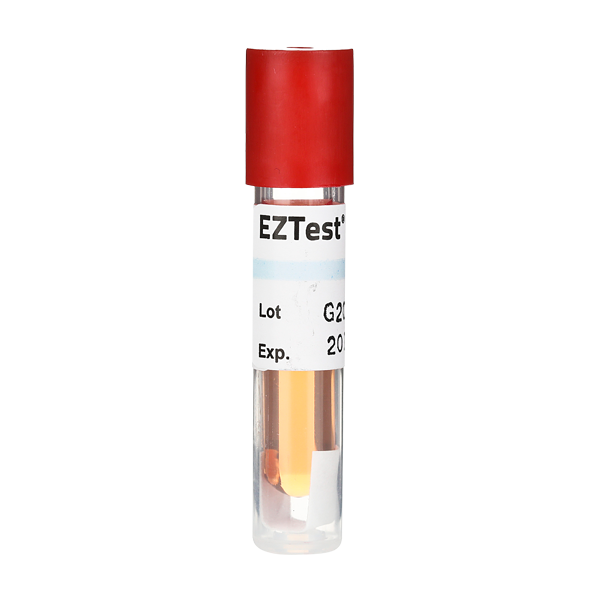
PCD® 2.13 Mesa EZTest EZG/6 SCBI (D-Value: 58)
PCD® 2.2 Steris BI Spore Strip, NA005 (D-Value: 28)
PCD® 2.5 Mesa BI Spore Strip in glassine, SGMG/6 (D-Value: 37)
PCD® 2.6 3M AttestTM 1264-S SCBI (D-Value: 58)
PCD® 2.7 Steris BI Spore Strip in glassine, NA005 (D-Value: 37)
PCD® 2.9 3M AttestTM 1294-S RRBI (D-Value: 58)
Type 3 (Least Resistance)
PCD® 3.13 Mesa EZTest EZG/6 SCBI (D-Value: 3)
PCD® 3.2 Steris BI Spore Strip, NA 005 (D-Value: 3)
PCD® 3.5 Mesa BI Spore Strip in Glassine, SGMG/6 (D-Value: 4)
PCD® 3.6 3M AttestTM 1264-S SCBI (D-Value: 5)
PCD® 3.9 3M AttestTM 1294-S RRBI (D-Value: 5)
Type 4 (Medium Resistance)
PCD® 4.1 Mesa BI Disc, 1-10006mm (D-Value: 21)
PCD 4.13 Mesa EZTest EZG/6 SCBI (D-Value: 50)
PCD® 4.2 Steris BI Spore Strip, NA005 (D-Value: 21)
PCD® 4.5 Mesa BI Spore Strip in Glassine, SGMG/6 (D-Value: 32)
PCD® 4.6 3M AttestTM 1264-S SCBI (D-Value: 50)
PCD® 4.7 Steris BI Spore Strip in Glassine, NA005 (D-Value: 32)
PCD® 4.9 3M AttestTM 1294-S RRBI (D-Value: 50)
Type 5 (Low Resistance)
PCD® 5.1 Mesa BI Disc, 1-10006mm (D-Value: 4)
PCD® 5.13 Mesa EZTest EZG/6 SCBI (D-Value: 13)
PCD® 5.2 Steris BI Spore Strip, NA005 (D-Value: 4)
PCD® 5.5 Mesa BI Spore Strip in Glassine, SGMG/6 (D-Value: 5)
PCD® 5.62 3M AttestTM 1264-S SCBI (D-Value: 13)
PCD® 5.92 3M AttestTM 1294-S RRBI (D-Value: 13)
Type 6 (Medium Resistance)
PCD® 6.1 Mesa BI Disc, 1-10006mm (D-Value: 12)
PCD® 6.13 Mesa EZTest EZG/6 SCBI (D-Value: 35)
PCD® 6.2 Steris BI Spore Strip, NA005 (D-Value: 12)
PCD® 6.5 Mesa BI Spore Strip in Glassine, SGMG/6 (D-Value: 17)
PCD® 6.6 3M AttestTM 1264-S SCBI (D-Value: 35)
PCD® 6.7 Steris BI Spore Strip in Glassine, NA005 (D-Value: 17)
PCD® 6.9 3M AttestTM 1294-S RRBI (D-Value: 35)
Type 7 (Medium Resistance)
PCD® 7.1 Mesa BI Disc, 1-10006mm (D-Value: 9)
PCD® 7.13 Mesa EZTest EZG/6 SCBI (D-Value: 26)
PCD® 7.2 Steris BI strip, NA005 (D-Value: 9)
PCD® 7.5 Mesa BI Spore Strip in Glassine, SGMG/6 (D-Value: 14)
PCD® 7.6 3M AttestTM 1264-S SCBI (D-Value: 26)
PCD® 7.7 Steris BI Spore Strip in Glassine, NA 005 (D-Value: 14)
PCD® 7.9 3M AttestTM 1294-S RRBI (D-Value: 26)
Type 8
Technical Specifications
Product Label: Polyolefin plastic with removable pressure sensitive test tube label.
Mounting Card: PVC plastic with a pressure sensitive adhesive strip for mounting on the outside of a sterilizer load.
EO Resistant Barrier Pouch: Proprietary multi-layer plastic film combinations result in consistent EO sterilization resistance to all commercial processes.
Biological Indicator: Per customer specification. Any commercially available BI test strip, disc or Self Contained BI (SCBI).
Related products
Related services

Contract studies laboratory
Mesa’s independent contract studies laboratory performs certified and compliant third-party chemical and biological indicator testing and verification.
Learn more

Sterilization cycle development
Get expert help for your bioburden. Mesa Labs assists with sterilization cycle development, BI qualification and validation, and routine monitoring support.
Learn more